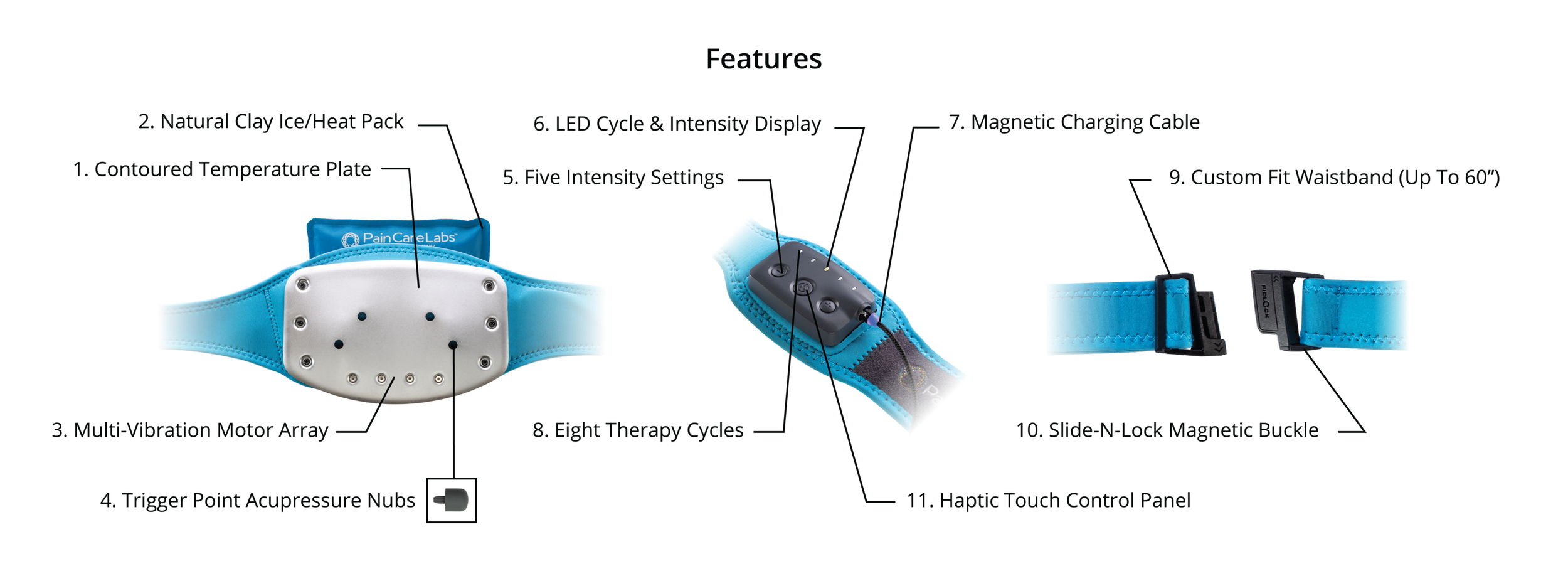
Reimagining the Brace: From Immobilizer to Active Therapeutic
By Amy Baxter, MD FAAP FACEP
This article was first published in Orthopedic Design & Technology Magazine.
The “movement is medicine” concept now pervades physical therapy. Motion speeds recovery, activates growth at a cellular level, and reduces pain by increasingly known and complex mechanisms. As the role of movement grows in medicine, how does immobilization fit?
Bracing reduces acute pain by restricting and protecting vulnerable anatomy from further damage, and can repair bone alignment over time. Yet in an era of accelerating neuromuscular, myofascial, and sensory‑modulation science, passive immobilization is increasingly inadequate. Both pro-movement and anti-immobilization studies suggest a benefit of cellular activation even when rigidity is required. Designers and manufacturers must now incorporate these discoveries to address the increasing likelihood that traditional bracing is often counter‑productive.
In this article, we explore how device manufacturers can adapt to this shift: why immobilization often fails or even harms; the physiology of injury in response to immobilization; how electrical and mechanical stimulation impact muscle and bone; how new “smart braces” can integrate stimulation, sensing, and adaptive control; and what design, regulatory and coverage implications emerge. The next generation of braces will be therapeutic devices in their own right, going beyond passive smart tracking to modulate tissue perfusion, osteogenesis, fascial mechanics, and pain, thereby improving long‑term functional outcome.
Immobilization
Brace design has historically emphasized rigid frames, straps, orthotic shells, and constraint. Images from the late 19th century conjure the dark side of bracing history: Elaborate contraptions forced on young children for genu valgum (knock knee) were later found to be largely unnecessary. For reforming bony defects such as club foot or scoliosis, sequential bracing is still clinically supported.
For injuries, however, the presumption that eliminating motion optimally supports healing structures (muscles, bones) may be inaccurate. Slight movement of multiple shoulder ligaments stimulates a humerus to regrow straight. Likewise, buckle fractures in children heal faster with bracing than casting.1 If the cellular activity of motion could be generated in everything from acute fractures to ligamentous disruption or post-operative fusion, there would be few true indications for immobilization. In addition, emerging evidence in musculoskeletal pain and dysfunction suggests that passive immobilization carries risk—especially where muscle, fascia, perfusion, or neuromotor control are involved.
Muscular hypoperfusion and deconditioning. Within days of any severe muscular, ligamentous, bony, or nerve injury, compensatory and supporting muscle derangement begins. The injured muscles undergo a pattern of inflammation, reactive hypertrophy, hypoperfusion, and spasm.2,3 External or pain-mediated immobilization leads to muscular fatty changes and further hypoperfusion within weeks,4,5 with inflammatory changes in fascia causing pain and instability as pain transitions to chronic.6-8 Reflex and proprioceptive responses to pain are altered, with this neuromotor hypofunction9 associated with ongoing functional instability and pain.10,11 Over time this leads to weak, fibrotic muscle that cannot sustain stabilization, contributing to the transition from acute to chronic pain.12
A brace that simply immobilizes will inadvertently reduce muscle strength in the stabilizers of the restricted plane. At worst, bracing can encourage patient passivity, decreased muscular engagement, fear avoidance, and eventual chronicity. In contrast, therapies that reduce pain and simulate motion may reduce the fear of moving while preserving muscle function. For developers of therapeutic braces, the challenge is no longer solely to provide structural support—it is to help maintain or restore tissue vitality, perfusion, neuromuscular control, and sensory integrity.
Sensors and Energy Stimulation In Bracing
A recent review of spinal bracing found 39 papers incorporated smart technology, ubiquitously in the context of accelerometers and monitoring.13 Likewise, a review of orthoses suggested that smart wearable orthoses could personalize correction and monitor movement.14 These uses could be amplified with incorporation of active bracing.
Tissues respond to energy stimulation, whether from electrical, ultrasound, pulsed electromagnetic fields (PEMF), vibrating plates, or focal mechanical sources. Electrical stimulation via transcutaneous electrical nerve stimulation (TENS) and focal vibration are well-established to reduce pain intensity, via central endogenous opioid release15,16 or neuromodulation, respectively. Studies pulsing electricity at amplitudes to cause muscular contraction show improved short-term patient-reported stability.17 Both whole body vibration and neuromuscular electrical stimulation (NMES) can increase strength.18 While focal mechanical stimulation (vibration) reversed hypotrophy after surgery, NMES did not (and was less well-tolerated).19,20 For bone growth, focal mechanical stimulation outperforms other energy sources, including ultrasound21 and PEMF.22 By matching the stimulation source and effective parameters to the tissue and goal, adding energy to immobilizers could enhance repair.
As organisms evolved to respond to mechanical force, incorporation of mechanical stimulation (M-Stim(r)) oriented to penetrate tissue is a logical active adjunct to smart bracing. The mechanism of mechanical force in cellular adaptation has only been recognized in the last decade. Ion channels are typically associated with electric and chemical charge for activation. Piezo1 channels are activated directly by pressure and held open with proteins using this force (e.g., Yoda1, Jedi1).24 Effects include musculoskeletal growth, vascular effects, and specific channels related to pain relief.24,25
These channels are reversibly opened, so intermittent pressure—vibration—can repeatedly activate cell activity, from reducing the lactic acid and LDH in post-exercise soreness to inhibiting and remodeling fatty lipids.26 In particular, vibration is highly anabolic for bone growth,23 with specific frequencies increasing blood flow, growth hormone expression, and reducing fatty tissue formation.
Bracing and Low Back Pain as a Model of Chronicity
One of the most highly reimbursed bracing targets is the low back. For chronic pain, rather than protecting the vertebrae and nerves, paraspinal muscles and thoracolumbar fascial derangement are increasingly viewed as targets for intervention.3,6,27
As with other trauma, muscles adjacent to the injured area hypertrophy, resulting in relative hypoperfusion and the same inflammatory ischemic cascade. Reduced blood flow contributes to persistent pain states, as the gliding myofascial layer retains its elastic state when hypoperfused, entrapping the nerve endings passing through.
Factors associated with non-specific low back pain (NSLBP) fit the paradigm of paraspinal muscle derangement pain, resulting most often from injuries associated with muscle strain and trauma (e.g., motor vehicle collisions).28 Prevention of chronic progression is associated with early muscle activation,29 physical therapy,30 increasing perfusion, and reducing inflammation and spasm.31,32
Chronic NSLBP reflects maladaptive healing and derangement of paraspinal muscle and fascia,6,33,34 including loss of normal microcirculatory function, impaired neuromuscular coordination, and central sensitization.8 These nonsurgical sources of pain explain why interventions aimed at structural correction (e.g., fusion) often fail, while early physical therapy and neuromodulation can modulate both local and central pain pathways to reduce dysfunction.
A recent pivotal randomized trial of the wearable multimodal brace DuoThermTM (Harmonic Scientific LLC, Delaware MD) supports this the paradigm shift. Funded by the NIH to replace opioids in patients with moderate‑to‑severe low‑back pain, the M-Stim device combined patient-controlled interactive thermomechanical frequencies transmitted through a rigid back plate. Frequencies targeting bone, muscle, vasodilation and pain neuromodulation were applied in a stochastic manner, with constructive interference patterns targeting different tissue planes.
After three or six months, the device prevented acute transition and reversed chronic low back pain dysfunction significantly better than the active control, TENS.35,36 For acute low-back pain users, only 31.8% in the M-Stim group progressed to chronic pain at three months vs 72.7% in the TENS group (RR 0.44, NNT 2.4). Importantly, first 28-day opioid use was reduced by 44.6% in the M-Stim group (32.33 morphine equivalents, p = 0.02), whereas opioid use increased in the TENS group.
The Active Brace: Design and Manufacturing Implications
For manufacturers of orthopedic devices and braces, the physics of mechanical stimulation matter: Frequency, interaction, amplitude, orientation, and thermal/pressure adjuncts target specific tissues. The braces of the future can be indicated to reduce transition from acute to chronic pain with early tissue-oriented therapy, while encouraging adherence by providing analgesia through multiple mechanisms.37
As vibration transmits best through solids, attachment of M-Stim units to rigid braces are an easy adaptation. A unit in vasodilatory frequencies could enhance DVT prophylaxis, or compensate for decreased perfusion with cold therapy compression while adding neuromodulatory pain reduction. Currently, device developers and scientists are stress-testing stimulation protocols: different frequencies support perfusion/muscle reactivation, neuromotor recalibration, spinal gating neuromodulation, and inhibition of inflammatory markers. Interaction of frequencies can yield different or broader results.
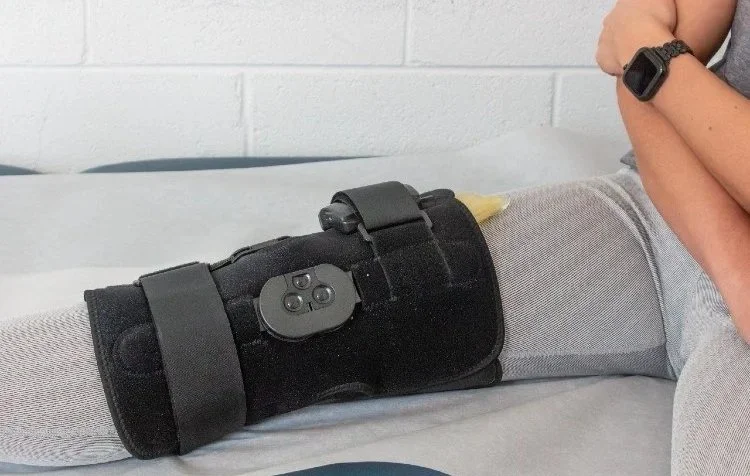
A patient uses VibraCool Pro for a knee injury.
Because a brace with embedded actuation is an active medical device, new FDA categories and HCPCS codes will be needed rather than simple orthosis: classification, safety (vibration exposure limits), biocompatibility, electrical safety, software IEC 62304, and cyber-security will all come into play. These aren’t just braces but therapeutic wearables, targeting pain/neuromuscular indications, post-op rehab, and prevention of chronic transition.
Read the rest of the article in Orthopedic Design & Technology Magazine.